|
The substance defats the skin, which may cause dryness or cracking. How is it bottom of the barrel diesel costs so much? It's the law! written by idiots and crooks. The chemical equation is:2 C10H22 31 O2 20) CO2 22 H2O What are the uses of decane - decane is a component of automotive fuels - decane is used in gas chromatography as standard. Molecular Formula, C10H22 or CH3(CH2)8CH3. "Sustainability" means you pay for it big time but don't have a right to receive it. Enviro-whinerism has carefully lowered the energy content/gallon. The most recently built refinery is ancient. Government intervention tortures your access to liquid fuels. $$\ce$-cut and up is cheap diesel and kerosene, with maximum cetane number. Cracking an object into smaller pieces to make it more useful is much like chemical cracking. We've got our compounds correctly expressed throughout all the equation, and now we need to move on to balance the equation. Sodium hydroxide plus hydrochloric acid yields sodium chloride and water. 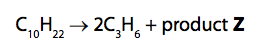
Cracking produces smaller molecules - alkanes, and alkenes. Let's hook them up and get the salt and water. Write the balanced equation for this reaction.  
If we have a long-chain hydrocarbon, such as decane, and we split it through thermal cracking (say in an industrial plant), we use high temperatures, and high pressures. ( 1 ) One molecule of the alkane decane ( C10H22 ) was cracked to give two molecules of propene and one.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
